1. Are Lithium-Ion Batteries Aqueous?
Most commercially available lithium-ion batteries are not aqueous batteries. They typically use organic carbonate-based electrolytes (such as EC/DMC), which have a wide electrochemical stability window (>4 V), enabling high energy density. However, these electrolytes are flammable and have poor thermal stability, posing safety risks.
In contrast, aqueous lithium-ion batteries (ALIBs) use water-based electrolytes. These systems are inherently safer, but their operating voltage window is limited due to water decomposition:
- Thermodynamic stability window: 1.23 V
- Actual electrochemical window: ~1.5–2.3 V
Due to these limitations, aqueous lithium batteries require carefully matched electrode materials and cannot directly reuse traditional lithium-ion battery materials.
2. A Practical Material System for Aqueous Lithium Batteries
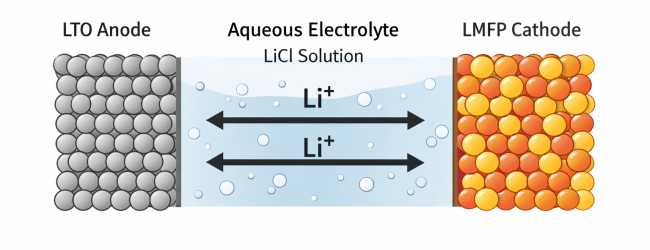
Instead of exploring complex material combinations, a practical and widely studied system consists of:
- Li₄Ti₅O₁₂ (LTO) – anode
- LiMnFePO₄ (LMFP) – cathode
- LiCl aqueous electrolyte – lithium-ion conductive medium
This system is commonly used in academic research, electrochemical testing platforms, and early-stage energy storage prototypes due to its balance of:
- Stability
- Safety
- Material availability
3. Core Material Properties and Electrochemical Parameters
3.1 Key Material Properties in Aqueous Lithium Battery Systems
To better understand the roles of each material, their key electrochemical properties are summarized below.
Lithium Titanate (Li₄Ti₅O₁₂, LTO) – Anode Material
- Working potential: ~1.55 V vs Li/Li⁺
- Theoretical capacity: 175 mAh/g
- Structural feature: Spinel structure with zero-strain characteristic (~0.2% volume change)
- Functional role: Provides highly stable lithium insertion with long cycle life
Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) – Cathode Material
- Working potential: ~3.4–3.6 V vs Li/Li⁺
- Theoretical capacity: ~170 mAh/g
- Structural feature: Olivine phosphate framework with strong P–O bonding
- Functional role: Enables stable lithium storage with reduced metal dissolution
Lithium Chloride (LiCl) – Aqueous Electrolyte
- Function: Lithium-ion conductive medium
- Solubility: High (~83 g/100 mL at 25°C)
- Structural feature: Highly soluble inorganic lithium salt
- Functional role: Provides Li⁺ transport and ensures safe, non-flammable operation
4. Lithium Titanate (Li₄Ti₅O₁₂, LTO): Structural Stability Enables Long-Life Aqueous Batteries

4.1 Electrochemical Characteristics
Lithium Titanate (Li₄Ti₅O₁₂, LTO) is widely recognized as a benchmark anode material in aqueous lithium battery systems due to its exceptional structural and electrochemical stability. It belongs to the spinel family and exhibits a highly stable three-dimensional lithium-ion diffusion framework.
LTO is often described as a “zero-strain” material, meaning its crystal lattice undergoes almost no structural distortion during lithium insertion and extraction. This characteristic is critical for maintaining long-term cycling stability.
Key electrochemical parameters include:
- Volume change: ~0.2% during cycling
- Flat voltage plateau: ~1.55 V vs Li/Li⁺
- Theoretical capacity: 175 mAh/g
- Crystal structure: Cubic spinel (Fd-3m space group)
4.2 Why LTO Is Ideal for Aqueous Systems
One of the key challenges in aqueous lithium batteries is avoiding hydrogen evolution reactions (HER) at the anode. This is where LTO provides a decisive advantage.
Because its working potential (~1.55 V vs Li/Li⁺) is significantly higher than that of graphite (~0.1 V vs Li/Li⁺), LTO operates safely within the electrochemical stability window of water.
This leads to several system-level benefits:
- Suppression of hydrogen evolution reactions in aqueous electrolytes
- Reduced parasitic side reactions, improving coulombic efficiency
- Enhanced electrochemical stability under repeated cycling
- Improved compatibility with aqueous lithium salts (e.g., LiCl)
In contrast, conventional graphite anodes are not suitable for aqueous systems due to their low operating potential, which triggers rapid water decomposition.
4.3 Cycle Life Performance
The structural robustness of LTO directly translates into excellent cycle life performance, which is one of its most important advantages in aqueous battery systems.
Experimental studies have demonstrated that:
- LTO-based aqueous cells can achieve >1000 stable charge–discharge cycles
- Under optimized conditions, cycle life can extend to several thousand cycles with minimal capacity decay
- Capacity retention remains high due to negligible structural degradation
This level of durability makes LTO particularly suitable for applications where long service life is more critical than maximum energy density.
4.4 Application Perspective
Due to its intrinsic safety and long-term stability, Lithium Titanate (Li₄Ti₅O₁₂, LTO) is widely used in various aqueous and hybrid battery systems.
Typical application scenarios include:
- High-safety energy storage systems
Especially in environments where thermal stability and reliability are critical - Fast-charging battery designs
LTO supports rapid lithium-ion insertion without significant structural stress - Aqueous lithium-ion battery research platforms
Frequently used as a standard anode material in academic and industrial R&D - Grid-scale and stationary storage systems
Where long cycle life (>1000–5000 cycles) is prioritized over energy density
From a materials engineering perspective, LTO is not just an anode choice—it is a stability anchor within aqueous lithium battery systems, enabling consistent performance under challenging electrochemical conditions.
5. Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP): A Balanced Cathode for Stability and Voltage

5.1 Structural and Electrochemical Features
Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) is a mixed transition metal olivine phosphate cathode material that combines the structural stability of iron phosphate with the higher voltage contribution from manganese.
LMFP belongs to the olivine phosphate family, which is well known for its strong polyanionic framework. The presence of PO₄³⁻ tetrahedra forms strong covalent P–O bonds, significantly enhancing structural rigidity and thermal stability.
Key electrochemical parameters include:
- Theoretical capacity: ~170 mAh/g
- Operating voltage: ~3.4–3.6 V vs Li/Li⁺
- Crystal structure: Orthorhombic olivine (Pnma space group)
- Redox mechanism: Fe²⁺/Fe³⁺ and Mn²⁺/Mn³⁺ couples
Compared to single-component systems such as LiFePO₄, LMFP offers a balanced combination of voltage and stability, making it particularly suitable for aqueous-compatible battery designs.
5.2 Stability in Aqueous Electrolytes
One of the primary challenges in aqueous lithium battery systems is the chemical stability of cathode materials, especially under repeated cycling in water-based electrolytes.
Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) demonstrates clear advantages over layered oxide cathodes (such as NCM or NCA):
- Lower transition metal dissolution in aqueous environments
- Stronger structural integrity due to the polyanion framework
- Reduced surface degradation reactions
- Improved long-term cycling stability
The stability originates from the inductive effect of the PO₄³⁻ group, which stabilizes the transition metal–oxygen bonds and suppresses oxygen release or structural collapse.
As a result, olivine phosphate materials like LMFP are widely considered more suitable for aqueous and hybrid electrolyte systems than conventional layered cathodes.
5.3 Application Perspective
Thanks to its balanced electrochemical performance and robust structure, Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) is widely used in both research and application-oriented battery systems.
Typical application scenarios include:
- Long-life energy storage systems
Where stable cycling performance is prioritized over peak energy density - Cost-sensitive battery applications
Due to the use of relatively abundant and less expensive raw materials - Safe cathode material development
Particularly in aqueous lithium-ion batteries and hybrid systems - Next-generation aqueous battery platforms
Where compatibility with water-based electrolytes is critical
From a system design perspective, LMFP serves as a reliable cathode counterpart to LTO anodes, enabling a stable and safe aqueous lithium battery configuration.
6. Lithium Chloride (LiCl) Aqueous Electrolyte: Ion Transport and System Stability
6.1 Physical and Chemical Properties

Lithium Chloride (LiCl) is a highly soluble inorganic lithium salt widely used in aqueous battery systems as an effective electrolyte component.
Due to its strong ionic nature, LiCl readily dissociates in water to form Li⁺ and Cl⁻ ions, enabling efficient ion transport within the electrolyte.
Key characteristics include:
- High solubility: ~83 g / 100 mL at 25°C
- Strong ionic dissociation: Rapid formation of Li⁺ carriers
- High ionic conductivity: Suitable for electrochemical systems
- Chemical stability: Stable under typical aqueous battery conditions
These properties make LiCl a practical and reliable choice for laboratory-scale and research-oriented aqueous lithium battery systems.
6.2 Electrochemical Role
In aqueous lithium-ion batteries, Lithium Chloride (LiCl) serves as the central medium for ion transport and system stability.
Its primary functions include:
- Enabling continuous Li⁺ ion transport between the cathode and anode
- Maintaining a stable ionic environment during charge–discharge processes
- Supporting electrochemical reactions at both electrodes
- Ensuring a non-flammable and safer electrolyte system compared to organic solvents
By providing a consistent supply of mobile lithium ions, LiCl plays a critical role in maintaining overall battery performance and efficiency.
6.3 Why LiCl Is Used in Research Systems
Compared to more complex electrolyte systems, Lithium Chloride (LiCl) is widely used in aqueous battery research due to its practical advantages:
- Simplicity: Easy to prepare and handle in aqueous solutions
- High reproducibility: Consistent performance across experiments
- Material compatibility: Works effectively with
- Lithium Titanate (Li₄Ti₅O₁₂, LTO) anodes
- Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) cathodes
- Cost-effectiveness: Readily available and suitable for scalable testing
These characteristics make LiCl an ideal electrolyte choice for:
- Electrochemical testing platforms
- Aqueous lithium battery prototyping
- Material performance evaluation
7. System-Level Integration: How LTO, LMFP, and LiCl Work Together
In a complete aqueous lithium battery system, these three materials function as a coordinated electrochemical unit:
- Lithium Titanate (Li₄Ti₅O₁₂, LTO) acts as the anode, providing stable lithium insertion with minimal structural change
- Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP) functions as the cathode, enabling reversible lithium storage
- Lithium Chloride (LiCl) serves as the electrolyte, facilitating Li⁺ transport between electrodes
System Operation Mechanism
- During charging: Li⁺ ions migrate from LMFP → LTO
- During discharging: Li⁺ ions return from LTO → LMFP
This coordinated interaction results in a system that is:
- Structurally stable
- Electrochemically reversible
- Safer than conventional organic electrolyte systems
8. Practical Applications of This Material System
The LTO + LMFP + LiCl system is not designed for maximum energy density, but for:
8.1 Key Advantages
- High safety (non-flammable electrolyte)
- Long cycle life (LTO stability)
- Low environmental impact
8.2 Application Scenarios
- Stationary energy storage systems
- Renewable energy integration (solar/wind buffering)
- Laboratory electrochemical testing
- Battery material research platforms
9. ULPMAT Full Aqueous Battery Material System
Building on the material framework of aqueous lithium batteries, ULPMAT provides a complete and integrated material platform, enabling users to move beyond single-material testing toward full system development.
Instead of sourcing individual components separately, this approach supports consistent material compatibility, simplified procurement, and faster experimental validation.
9.1 Material Coverage
ULPMAT’s aqueous battery material system includes a comprehensive range of functional materials across all key components:
- Anode materials:
Including Lithium Titanate (Li₄Ti₅O₁₂, LTO) and other titanium-based insertion compounds designed for high structural stability and long cycle life - Cathode materials:
Including Lithium Manganese Iron Phosphate (LiMnFePO₄, LMFP), Lithium Iron Phosphate (LiFePO₄, LFP), and related olivine phosphate materials suitable for aqueous-compatible systems - Electrolyte materials:
Including Lithium Chloride (LiCl) and other lithium salts used for aqueous electrolyte formulation and electrochemical testing - Extended aqueous systems:
Supporting materials for sodium-based aqueous batteries, such as NASICON-type compounds and Prussian blue analogs
This structured material coverage allows users to design, test, and optimize aqueous battery systems within a unified material framework.
9.2 System Advantages
By offering a full material system rather than isolated products, ULPMAT enables several practical advantages:
- Faster experimental setup
All key materials are available within a coordinated system, reducing sourcing complexity and setup time - Consistent material compatibility
Materials are aligned in terms of electrochemical behavior, improving system stability and reproducibility - Scalable transition from research to application
Suitable for both laboratory-scale testing and early-stage application development - Improved development efficiency
Reduces trial-and-error in material selection, allowing more focus on system optimization
10. FAQ: Lithium-Based Aqueous Battery Materials
Q1. Are aqueous lithium-ion batteries safer than traditional lithium-ion batteries?
Yes. Aqueous lithium-ion batteries use water-based electrolytes, which are non-flammable and significantly reduce the risk of thermal runaway compared to organic electrolyte systems.
Q2. Why is LTO preferred over graphite in aqueous battery systems?
LTO operates at a higher potential (~1.55 V vs Li/Li⁺), which helps avoid hydrogen evolution reactions in aqueous electrolytes. In contrast, graphite operates at much lower potentials and is unstable in water-based systems.
Q3. What makes LMFP suitable for aqueous cathodes?
LMFP has a stable olivine structure with strong P–O bonds, which reduces transition metal dissolution in aqueous environments and improves long-term cycling stability.
Q4. Can LiCl be replaced with other lithium salts in aqueous batteries?
Yes. Other lithium salts (such as Li₂SO₄ or LiNO₃) can also be used. However, LiCl is widely chosen for its high solubility, strong ionic conductivity, and experimental simplicity, making it suitable for research and prototyping.
Q5. What are the main applications of aqueous lithium battery systems?
Aqueous lithium battery systems are mainly used in:
- Stationary energy storage
- Renewable energy buffering systems
- Laboratory-scale battery research
- Safe battery prototyping
These applications benefit more from safety and cycle life than from maximum energy density.
11. Conclusion
While most lithium-ion batteries are not aqueous, aqueous lithium systems represent a clear direction toward safer and more sustainable energy storage.
Among many material combinations, the system based on:
- LTO (Li₄Ti₅O₁₂)
- LMFP (LiMnFePO₄)
- LiCl electrolyte
offers a practical, research-supported, and scalable solution.
With increasing demand for safety and sustainability, this material system is becoming an important platform for next-generation battery development and research.