What Is an Aqueous Battery and Why It Matters in Energy Storage?
An aqueous battery is a secondary battery system that uses an aqueous solution as the electrolyte. Its core characteristic lies in utilizing water as the ion-conducting medium, giving it significant advantages over organic electrolyte systems in terms of safety, cost, and environmental friendliness. Compared to traditional non-aqueous batteries, aqueous batteries naturally possess characteristics such as non-flammability, high ionic conductivity, and strong system stability, thus holding significant application potential in large-scale energy storage, low-cost electrochemical energy storage, and safe battery systems.
What Are Phosphate Cathodes and NASICON Anodes in Aqueous Sodium-Ion Batteries?
In current aqueous battery material systems, phosphate-based cathodes and NASICON-structured anodes have become the most widely studied and practically relevant choices for aqueous sodium-ion batteries. Among them, phosphate materials—especially NaFeMnPO₄-type systems—are highly valued due to their stable crystal structure and predictable electrochemical behavior in aqueous environments. Their primary function is to provide a structurally robust cathode platform that can maintain integrity during repeated sodium-ion insertion and extraction processes.
At the same time, NASICON-type materials such as NaTi(PO₄)₃ are widely regarded as promising anode candidates. Their three-dimensional ion diffusion channels enable fast and reversible sodium-ion transport while maintaining excellent structural stability, which is essential for long-cycle operation in aqueous systems.
From a practical engineering perspective, these two material families address the two most critical requirements of aqueous sodium-ion batteries: structural safety and ion transport efficiency. Phosphate-based cathodes offer strong chemical and mechanical stability in highly polar electrolytes, while NASICON anodes provide fast reaction kinetics and long-term cycling reversibility. This complementary behavior is a key reason why they are currently considered one of the most promising material combinations for aqueous sodium-ion battery development.
At ULPMAT, the current material portfolio is mainly focused on three core directions: phosphate-structured cathode materials, NASICON-structured sodium-ion anode materials, and related supporting materials designed to optimize overall system performance and interface stability.
What Is NaFeMnPO₄ Phosphate Cathode Material and How Does It Work in Aqueous Sodium-Ion Batteries?
NaFeMnPO₄ is a structurally stable phosphate-based cathode material designed for aqueous sodium-ion batteries, where its PO₄ framework provides inherent resistance to structural degradation and hydrolysis in water-based electrolytes.
NaFeMnPO₄, as a phosphate-based cathode material, achieves its functionality primarily through its stable PO₄ tetrahedral crystal framework and the synergistic structure of the Fe–Mn dual transition metals. During electrochemical processes, Fe²⁺/Fe³⁺ provides the main reversible intercalation/deintercalation reaction pathways, ensuring the stability of the basic capacity output, while Mn²⁺/Mn³⁺ participates in reaction regulation across different potential ranges, thereby expanding the overall voltage window and enhancing the system’s reaction tolerance. This dual red oxygen center design allows the material to maintain low structural stress concentration in aqueous or highly polar electrolyte environments, resulting in more stable cycle performance.
In terms of material preparation, Sodium Iron Manganese Phosphate (NaFeMnPO₄) is typically prepared using a wet chemical precursor method combined with high-temperature solid-state sintering. First, elemental uniformity is achieved through solution blending or co-precipitation of Fe and Mn sources. Then, a phosphate precursor is introduced to construct the initial framework structure, followed by drying and high-temperature calcination to form a stable phosphate crystal phase. To further enhance performance, industrial-scale preparation typically incorporates carbon coating to improve electronic conductivity, and optimizes crystal integrity and ion diffusion paths through particle size control and atmosphere management, thereby achieving controllable engineering production of the material.
In terms of applications, Sodium Iron Manganese Phosphate (NaFeMnPO₄) is currently primarily in the engineering verification and application introduction stage for aqueous sodium-ion batteries and safe energy storage systems, widely used in the research and development of low-cost, large-scale energy storage battery cathode materials. Simultaneously, this material is also used in material screening platforms for next-generation safe energy storage systems to evaluate long-cycle stability and structural durability in aqueous systems. Due to its combination of structural stability and tunable electrochemical performance, NaFeMnPO₄ is considered one of the important candidate materials with engineering potential for future aqueous sodium-ion battery cathode systems.

What Is NaTi(PO₄)₃ NASICON Anode Material and How Does It Work in Aqueous Sodium-Ion Batteries?
NaTi(PO₄)₃ is a NASICON-structured anode material designed for aqueous sodium-ion batteries, where its three-dimensional open framework enables fast and reversible sodium-ion transport while maintaining excellent structural stability in water-based electrolyte environments.
NaTi(PO₄)₃, as a NASICON-type anode material, achieves its functionality primarily through its three-dimensional ion-conducting crystal framework and the stable Ti-based redox center. During electrochemical processes, the Ti⁴⁺/Ti³⁺ redox couple provides a highly reversible insertion/extraction reaction pathway, ensuring stable charge storage behavior, while the NASICON structure offers interconnected ion diffusion channels that significantly enhance sodium-ion mobility. This open framework design allows the material to maintain low structural distortion during repeated cycling, effectively reducing stress accumulation and improving long-term electrochemical stability in aqueous or highly polar electrolyte systems.
In terms of material preparation, Sodium Titanium Phosphate (NaTi(PO₄)₃) is typically synthesized through a solid-state reaction route or a sol–gel assisted high-temperature calcination process. First, titanium and sodium precursors are uniformly mixed with phosphate sources to form a homogeneous precursor system. This is followed by controlled drying and high-temperature sintering to induce the formation of a stable NASICON crystal phase. To further improve electrochemical performance, industrial preparation often incorporates particle size optimization, morphology control, and carbon coating strategies to enhance electronic conductivity and accelerate ion transport kinetics, enabling more reliable and scalable material production.
In terms of applications, NaTi(PO₄)₃ is currently mainly used as a key anode material in aqueous sodium-ion battery systems and advanced safe energy storage technologies. It is widely applied in laboratory-scale cell validation, full-cell configuration studies, and long-cycle stability evaluation platforms for water-based battery systems. Due to its combination of structural robustness, fast ion transport capability, and intrinsic safety advantages (no metal plating behavior), NaTi(PO₄)₃ is considered one of the most promising anode candidates for next-generation aqueous sodium-ion battery systems requiring high safety and long operational lifetime.

LiNbO₃ Coating and LiCl Electrolyte Additive in Aqueous Sodium-Ion Batteries: Interfacial Engineering Roles and Function
In aqueous battery systems, performance depends not only on the electrode materials themselves but also directly on the interfacial state between the electrode and the electrolyte. In actual operation, many capacity decay and cycle instability issues do not originate from the material itself but from interfacial side reactions, water molecule erosion, and the gradual destabilization of the surface structure. Therefore, interface control plays a crucial engineering role in aqueous batteries.
LiNbO₃: Protective Coating Material for Electrode Surfaces
In practical applications, LiNbO₃ is typically used as a coating layer on the electrode material surface to improve its stability in aqueous environments. Its main function is not to participate in electrochemical reactions but to act as a “protective interface,” reducing direct contact between the electrolyte and the active material.
Specifically, this coating layer can:
- reduce the possibility of water molecules directly penetrating the electrode crystal;
- reduce the dissolution of active metal ions and structural damage;
- form a relatively stable interfacial protective layer on the electrode surface, mitigating side reactions;
- improve structural retention during long-term cycling;
From an engineering perspective, LiNbO₃ is more like an “interfacial isolation membrane,” used to extend the lifespan of electrode materials in aqueous environments.
LiCl: A Structure-Regulating Salt for Electrolyte Systems
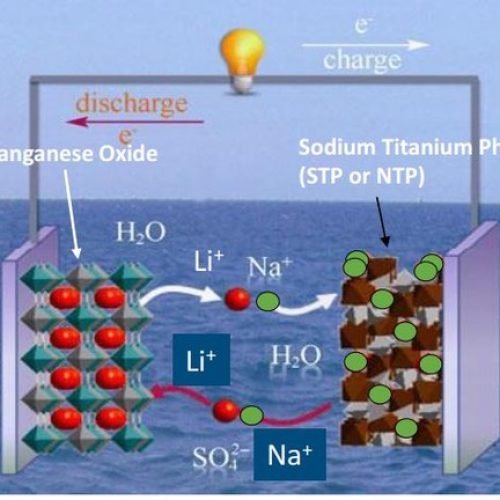
In high-concentration aqueous electrolyte systems, LiCl’s role is not only to provide ionic conductivity, but more importantly, to participate in regulating the overall microstructure of the electrolyte, thereby improving the battery’s operating environment.
In practical systems, it mainly plays the following roles:
- Adjusting the arrangement of water molecules, reducing the proportion of “free water”Changing the solvation structure around ions, making ion migration behavior more stable
- Extending the stable operating range of the electrolyte under high voltage conditions
- Reducing the probability of side reactions and improving overall cycle stability
From an application perspective, LiCl is more like an “electrolyte environment modifier,” indirectly improving the overall performance of the battery by changing the distribution of ions and water structures within the aqueous system.
summary
In general, aqueous sodium-ion batteries rely on integrated material systems rather than single components. Phosphate-based cathode materials provide structural stability and reliable redox performance in aqueous environments, while NASICON-structured anode materials enable rapid and reversible sodium-ion transport through their three-dimensional ion channels, supporting long cycle life and high safety. Furthermore, interface and electrolyte engineering materials further enhance battery stability by suppressing side reactions and optimizing the solvation structure of the electrolyte.
The ULPMAT material portfolio covers the following main categories in this system:
- Phosphate-based cathode materials
- NASICON-structured anode materials
- Interface coating materials for electrode protection
- Electrolyte additives and solvation structure modifiers
- Auxiliary functional materials for full-cell system optimization
Contact us for a product list